Investigation of Causality by the Philippines Covid-19 Pharmacovigilance System as of April 2023: 38% of the reported deaths were investigated with 8 causally linked. 1.8% of reported AEFI were
investigated with 323 cases found to be causally linked. 0.8% of cardiac AESI were investigated, with no causality findings stated. The system is spontaneous (passive) and will be underreporting.
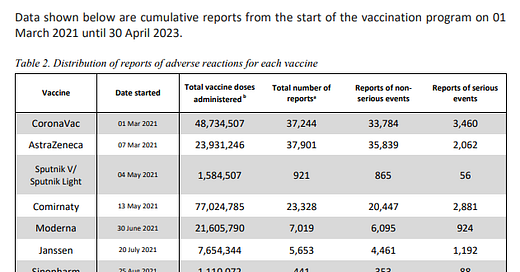
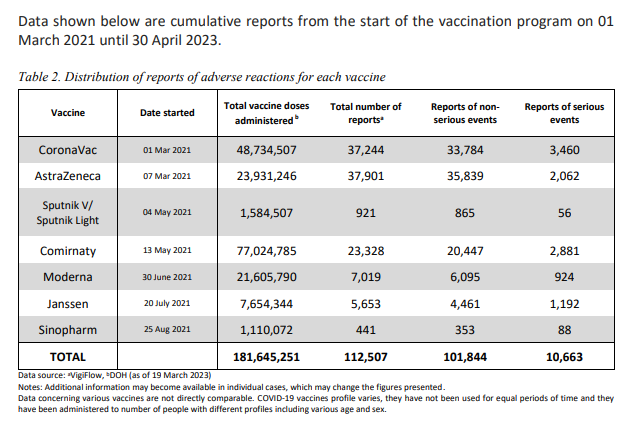
This is the reports of adverse events from the April 30, 2023 Philippines Pharmacovigilance report. Deaths are not shown in Table 2, but are shown in the text in another part of the report.
FOI request regarding investigation of causality was made, and response was received. The following questions were asked and responses were received. My comments are in bold and italics.
Q1. of the 2,816 deaths, how many have
- been subject to root cause analyses
- found to be product related
- arisen from which productResponse:
Based on spontaneous reporting system (voluntary submission of reports of adverse events following immunization), there were 2,816 reports with fatal
outcome. About 1,075 cases have been subjected to causality assessment.
Most cases are coincidental events.
Eight (8) of the reports were found to be vaccine product related reactions (caused or precipitated by a vaccine due to one or more of the inherent properties of the vaccine product). These include 2 cases using Vaxzevria, 2
Jcovden, 3 Comirnaty, and 1 Sinovac. 1,075 / 2,816 = 38% of the deaths were subject to causality assessment, resulting in findings that 8 deaths were caused or precipitated by a vaccine. If all of the deaths had been evaluated and causality were assessed in the same proportions this would be equivalent to 21 deaths.
What now if the underreporting factor were 40? That could mean 838 deaths.
What if it were 100? That could mean 2,100 deaths?
What if it were 1000? That could mean 20,000 deaths, and doesn’t explain the now well more than 300,000 excess deaths observed in the Philippines since the start of rollout of Covid-19 vaccines in March of 2021 and up to end of 2022, in still preliminary vital statistics data.
How many causally linked deaths might be a concern?
Q2. of the 112,507 reports of AEFI, how many have
- been subject to root cause analyses
- been found to be product relatedResponse:
Of the total reports, 2,112 cases were subjected to causality assessment. Using the WHO causality assessment classification, most cases were classified as C. Coincidental while 323 were classified as A. Consistent with causal association to immunization.2,112/112,507 = 1.9% of the total reports were subject to causality assessment, resulting in 323 meeting causality criteria. If all of the cases had been evaluated and causality were assessed in the same proportions this would be equivalent to 17,206 adverse events causally linked.
What now if the underreporting factor were 40? That could mean 688,253 adverse events.
What if it were 100? That could mean 1.7 million adverse events?
What if it were 1000? That could mean 17 million adverse events out of 181 million vaccine doses?
COULD MEAN - there is simply not enough data to draw conclusions.
How many adverse events might be a concern?
Q3. Does the FDA depend on the reporting doctor / party for determination of cause of death, including identification of "adverse events of special interest" (AESI), or are these independently analyzed? Response:
The FDA relies on the information provided by the
reporter. In the science of pharmacovigilance, it is called
spontaneous reporting. In some instances, the Department of Health through the Epidemiology Surveillance Units conducts investigation to further
gather information on cases needing additional information necessary to conduct causality assessment by regional/national adverse events following immunization committee.This pharmacovigilance system is spontaneous and passive. It relies on the reporter to provide information. If the reporter does not / cannot provide much data or evidence on the reported injury or death, then there will be no prospect for a causality assessment to proceed.
Passive reporting systems are expected to grossly under capture and under report adverse events. The FDA does note that “in some instances”, the DOH conducts further data gathering.
Cardiac Symptoms
There is a breakdown of symptoms by vaccine type. I tallied all the cardiac symptoms and asked how many had been investigated.
Q4. Of the 2,822 Cardiac Symptoms, how many have been reviewed for possibility of being due to an AESI?Response:
According to Brighton Collaboration, adverse events of special interest include myocarditis/pericarditis and other forms of acute cardiac injury including arrhythmias, heart failure, coronary artery disease, myocardial infarction,
and stress cardiomyopathy. 24 cases of AESI have been subjected to causality assessment.Apparently 24 out of 2,822 cases of cardiac symptoms = 0.8% of received reports, have been investigated. No statement on causality outcomes was provided in the response.
The response ends with stating FDA’s consistent stance since the first pharmacovigilance report in early 2021; that the report only assesses the overall benefit-risk, not on individual risks. That benefits outweigh risks.
Never mind that population risks are comprised of individual risks.
Note that the Pharmacovigilance Report assess the overall benefit risk of the COVID-19 vaccines and not on the individual risks. Based on the reporting rate of 0.06%, data suggests that the benefits of using COVID-19
vaccines far outweigh its risks.Below was the pharmacovigilance budget as informed last September 2022, which was around 40 million from 2020 to 2022. The 2023 budget is not stated.
Is the Pharmacovigilance system fit for purpose? Has it adequately captured adverse events? Is it able to capture and raise any alarm?
Is it time for an investigation? Please share and demand accountability from health officials and government.