NAEFIC Head twice declines to attend the ongoing Philippines Excess deaths hearing. Did the Philippines Covid-19 Pharmacovigilance System Fail Filipinos?
Only 52 reactions of special interest, none confirmed fatal, are recognized by FDA. I provide a list of questions to address the adequacy of NAEFIC to assess the 113,166 adverse reactions reported.
The purpose of the NAEFIC was to assess causality of reports of adverse reactions in the Philippines following Covid-19 immunization.
At last week’s senate hearing on 11th June 2024, there was a discussion surrounding the NAEFIC committee and why the head of this committee had declined to attend the hearing despite repeated invitations. Dra. Villaneuva is a private clinician who was appointed to head this committee. In response to questioning, the DOH advised that all persons on the committee were experts appointed from outside the DOH, non-government personal, to allow non-partial, non-biased assessment of the AEFI cases.
We must note the following statement from 21 April 2021, about 7 weeks following the start of the Covid-19 vaccines rollout, where a member of NAEFIC said that the committee was looking for clustering of adverse events and would not hesitate to halt the rollout if it that were warranted. I somehow wonder how 113,166 cases, 10,805 serious with 2,864 deaths never managed to constitute a cluster; particularly when most of the reported deaths (as evidenced from my prior assessment of the PH VAERS reports occurred within 0 - 3 days post injection).
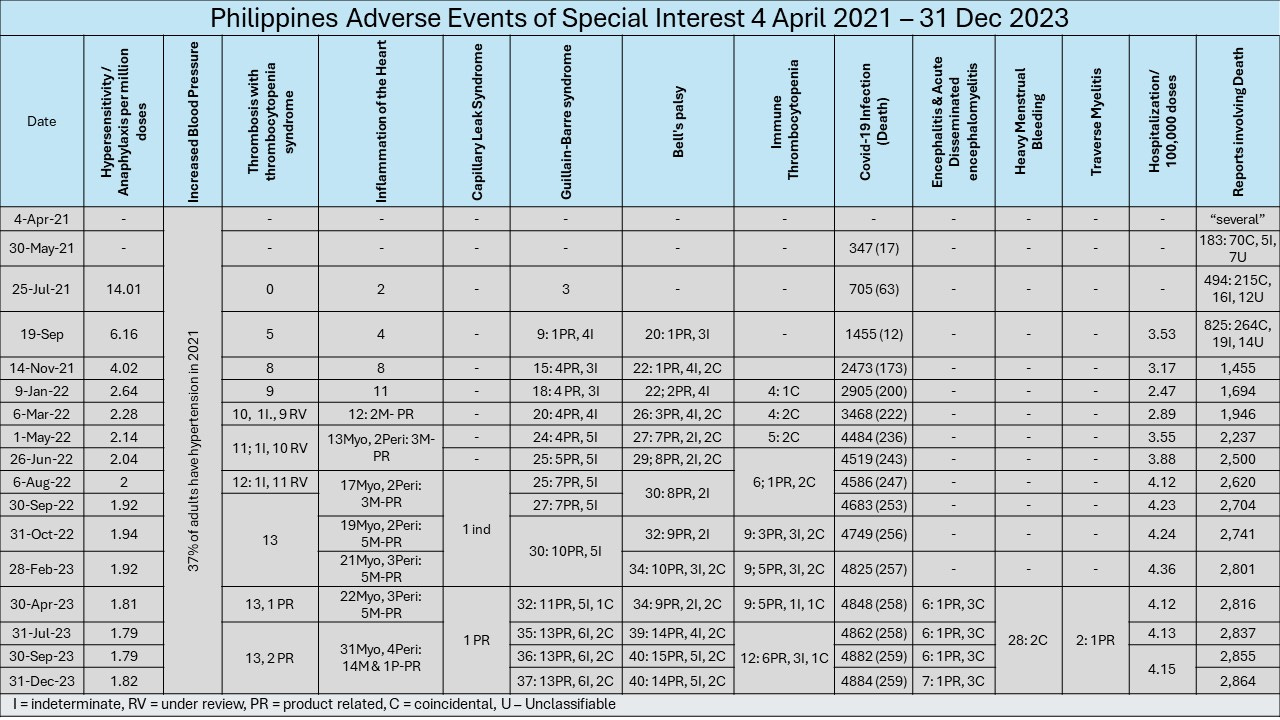
The FDA started publishing Pharmacovigilance reports presenting outcomes following immunization on 4th of April 2021, 1 month after the start of rollout. This was later updated with reports starting on 7th March 2021. Note that the FDA report dated 18th April 2021, 3 days prior to the above published NAEFIC statement, showed the delivery of 1,279,223 doses of vaccines and reports of 26,831 adverse reactions, including 401 serious reactions and “several” deaths.
At the time of the final FDA Report on 31 December 2023, 181,645,251 doses had been delivered up to March 2023 (there was still rollout of vaccines and boosters later than March 2023, but these are not reflected in the FDA reports or any DOH official public domain data), with reports of 113,166 AEFI, 10,805 of them serious including 2,864 deaths.
The 31 December 2023 final report contained ONLY the following 52 incidents of recognized adverse events of special interest out of the 113,166 reports made to the FDA.
Thromosis-Thromobcytopenia Syndrome:
2 cases of thrombosis-thrombocytopenia were reported and are considered product linked. 13 cases of thrombosis were under review.
Confirmed Covid-19 Infections:
There were 4,884 confirmed reports, mostly asymptomatic and with 259 fatal outcomes. Cases were considered unrelated to vaccination and were actual natural infections.
Inflammation of the Heart:
14 cases of myocarditis (out of 31 reported cases) and 1 case of pericarditis (out of 4 reported cases) were considered product linked. The remaining 17 cases of myocarditis and 3 cases of pericarditis were still under review.
Capillary Leak Syndrome:
1 case of this syndrome was reported and was considered product linked.
Guillain-Barre Syndrome:
13 cases out of 37 cases reported were considered product linked, 6 were judged indeterminate, 2 coincidental, and 16 under review.
Bell’s Palsy
14 cases out of 40 cases reported were considered product linked, 5 were judged indeterminate, 2 coincidental, and 19 under review
Immune Thrombocytopenia
6 cases out of 12 cases reported were considered product linked, 3 were judged indeterminate, 1 coincidental, and 2 under review
Encephalitis and Acute Disseminated Encephalomyelitis
1 case out of 7 cases reported were considered product linked, 3 coincidental, and 3 under review.
Heavy Menstrual Bleeding
28 cases reported, 9 being serious. 2 were judged as coincidental and the rest have been under review since 30 April 2023.
Transverse Myelitis
1 out of 2 cases reported was assessed as product related and the other is under review
Hospitalization Rate was 4.15 per 100,000 doses administered. None of these are acknowledged as being vaccine related.
Fatal Events
2,864 fatal events reported. None of these are acknowledged as being vaccine related.
I monitored these recognized adverse events and there was literally no movement on the cases under review for months, raising the question as to the adequacy of NAEFIC to identify, evaluate and act on adverse reactions. There are only 52 acknowledged product-related AESI (adverse events of special interest) out of 113,166 reports, 10,805 of them serious for 181 million doses of various products! I am not buying this!
Further, the rates of reporting of adverse events dropped off very sharply after the first few months of rollout, even as there was massive ramping up of dosing. Was there active dis-incentivization for reporting of adverse events, even aside from ubiquitous the Safe and Effective mantra that was continuously marketed to the naive and trusting public?
Questions for the NAEFIC
How many persons on the committee & what is their background? Were there any changes in committee members from first hearing to last?
When did this committee start meeting, date?
When did it finish meeting / are there still ongoing meetings?
How often did this committee meet? How many total hours did the committee meet (dates and durations)? Attendance record for each of the committee meetings?
Who prepared the reports that the committee deliberated upon? How did they select AEFI/SAE reports for review?
How many cases were reviewed? How many cases were concluded?
Were the 52 concluded cases automatically referred to Philhealth for compensation (considering that causality was concluded by the committee?)
What was the checklist of requirements / flowchart for determination of causality?
I hope that someone is watching these posts and will make reference to these at the next hearing. The failures of the Pharmacovigilance system must not be repeated in case of any future pandemic situation.



















Perhaps the question that should be asked of all NAEFIC committee members under penalty of law, is whether or not they have received ANY financial compensation or compensation in kind by any of the pharmaceutical companies of which they are supposed to have oversight. "Show me the incentive and I will show you the outcome" Charlie Munger
" For the love of money is a root of all kinds of evils, for which many have strayed from the faith in their greediness, and pierced themselves through with many sorrows." 1Timothy 6:10 Be....in the Word, the Truth sets us free.
If the system in place is anything like the one in The US, called VAERS (Vaccine Adverse Event Reporting System), where it is known and acknowledged that only 1 to 10% of all adverse events are reported, then you can imagine how many more cases of adverse reactions there have been and will continue to be. The boosters make the problems even worse. In the US the incentives were to the tune of an extra 40,000 dllrs for vaccinating 100 children in some cases.