Philippines Pharmacovigilance: Review of FDA Reports and VAERS Reports for Persons Aged 40 to 59.
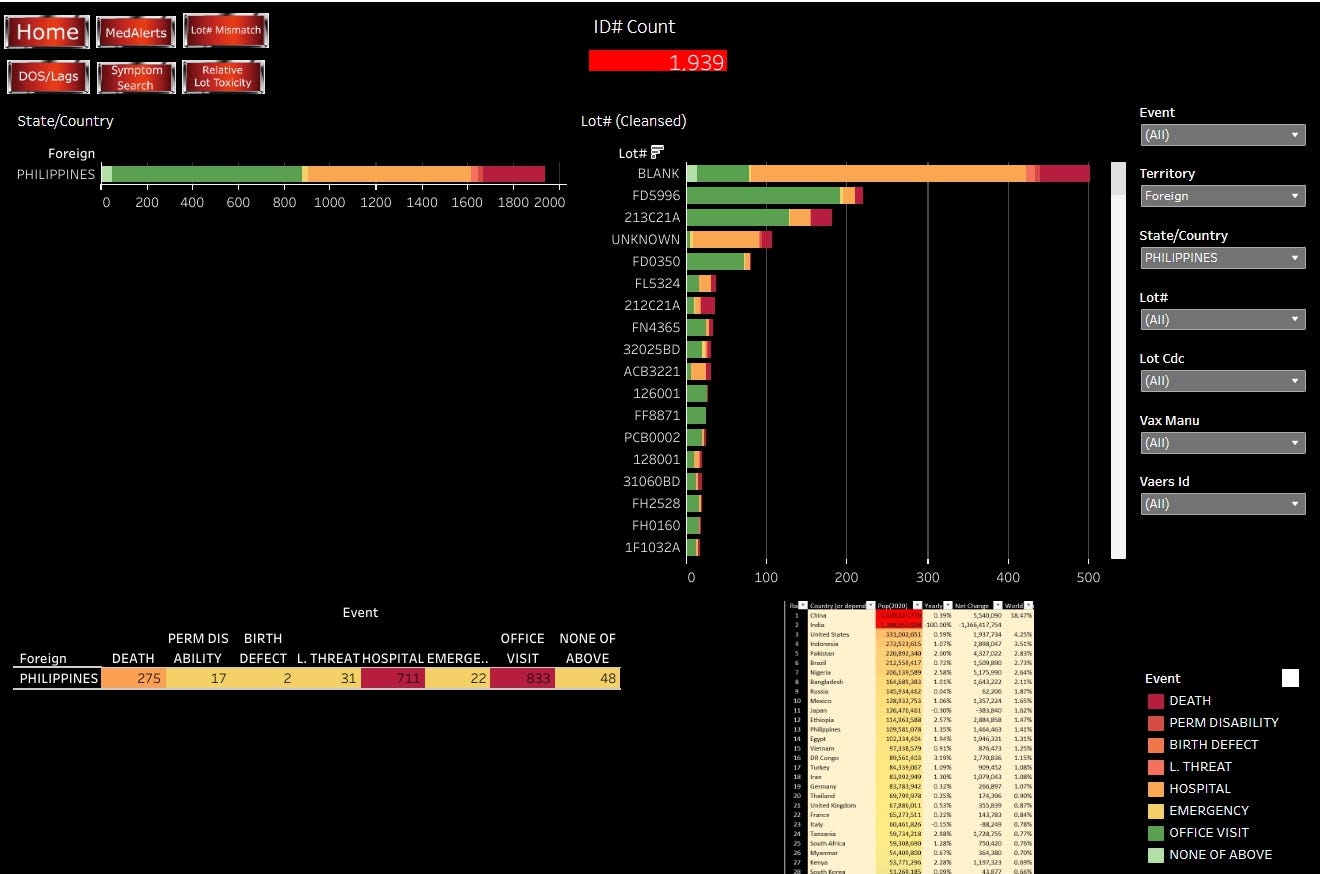
Philippines FDA reported 793 deaths for this age-group, while VAERs reported 1,939 ID counts of SAE with 275 Deaths. By Batch, Janssen had the most reports of deaths and SAE, attributed to 2 batches.
Persons aged 40 - 59 are in the peak of their professional lives, even though some may begin to experience health challenges at this time. Overall risks from Covid-19 infections were generally considered to be low for most persons in this age bracket.
The Philippines FDA has received reports of 793 deaths suspected to be AEFI for persons aged 40 to 59.
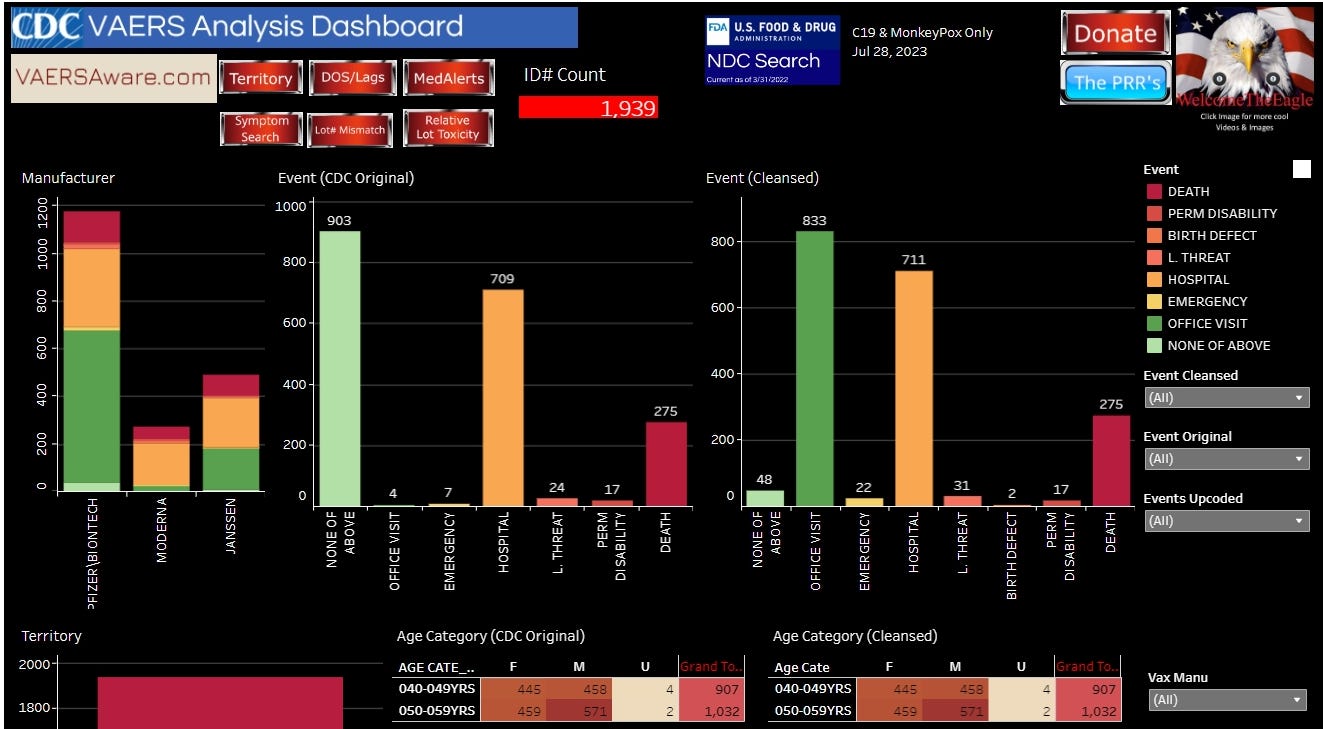
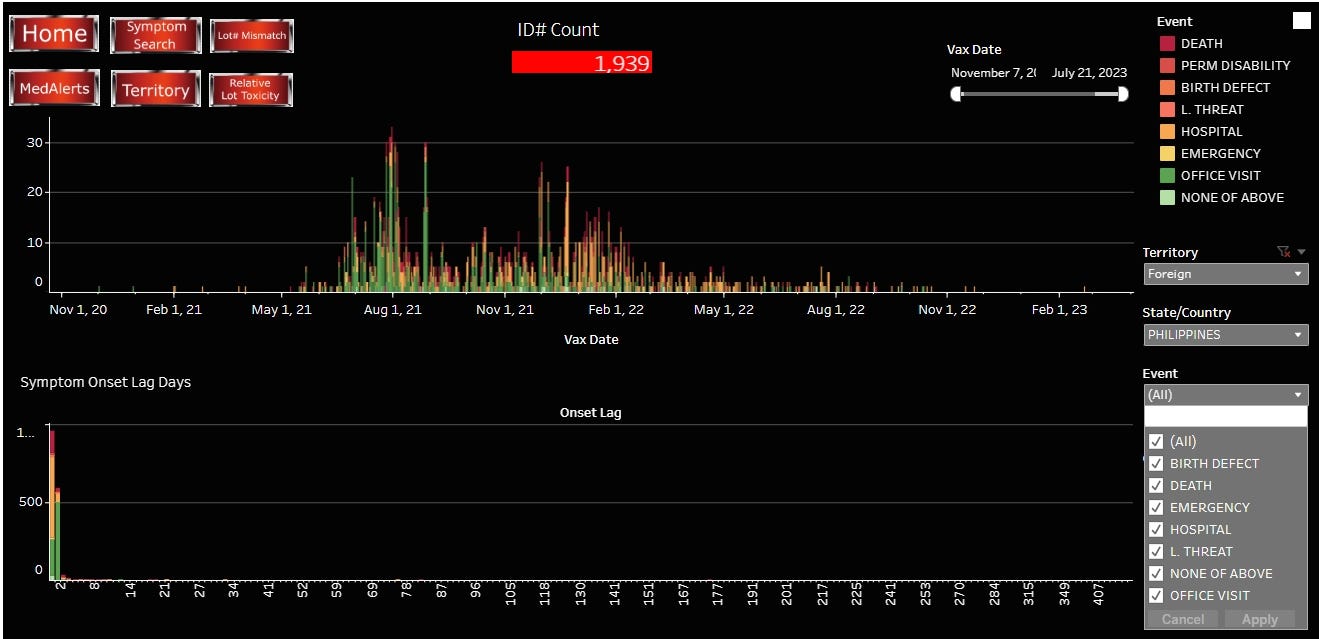
The VAERSAWARE SYSTEM reports 1,939 ID Counts from the Philippines. 275 deaths, 17 permanent disabilities, 2 birth defects, 3 life-threating events, 711 hospitalizations, 22 emergency room visits, 833 Dr office visits and 48 “none of above”.
By manufacturer, there were 131 deaths reported following Pfizer, 54 deaths following Moderna, and 90 deaths following Janssen. Janssen deaths are disproportionate given the relatively lower dosing for this product.
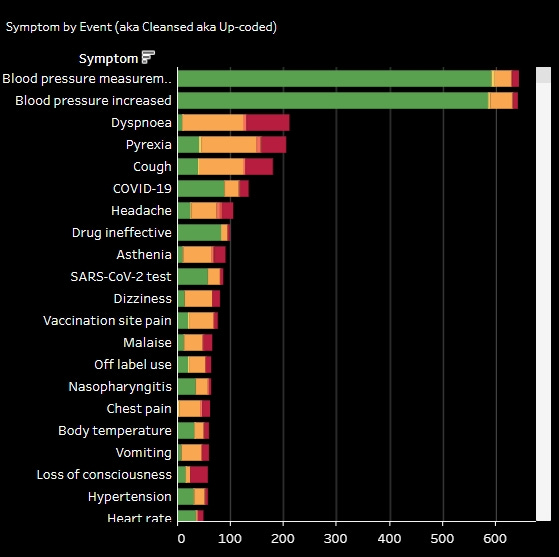
The overwhelmingly most common symptoms reported was increased blood pressure, followed by shortness of breath, fever, and cough.
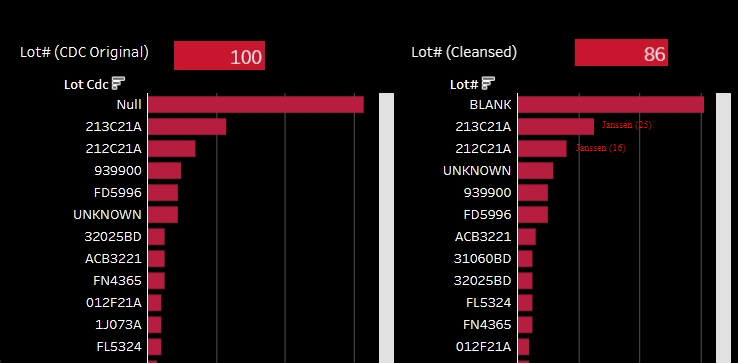
In terms of deaths, the two Janssen Batches 212C21A and 213C21A were responsible for the most reported deaths. Even though, Janssen dosing (3.2 million for the delivery including these batches) was overall much lower than both Pfizer (overall 77 million) and Moderna (overall 21.6 million) dosing. Further there will likely be many Janssen doses hidden in the blank and unknown reports.
Onset of SAE is very much concentrated around days 0 - 1 following injection.
Selected adverse events reports: -
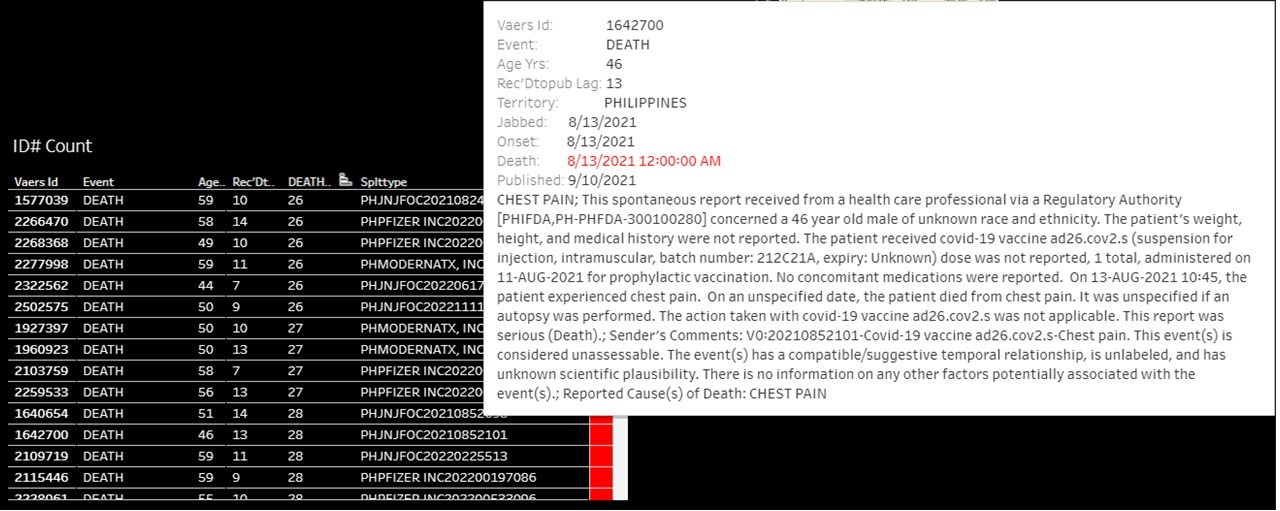
Death in a 46-yo-male with key symptom of chest pain 2 days after he received a dose of Janssen 212C21A. Report is considered unassessable (due to limited information), though compatible temporal relationship is acknowledged.
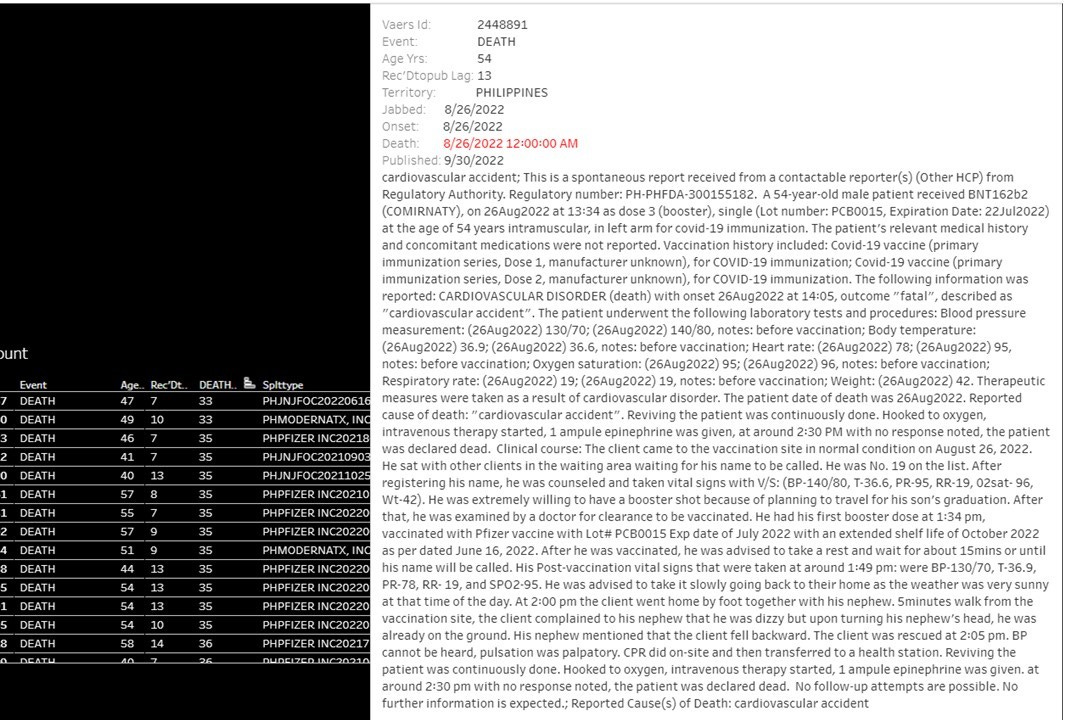
Death from heart attack in a 54-yo-male the same day that he received a 3rd booster dose of Comirnaty. He had presented to the vaccination center in normal health, died as he was exiting the center to go home, and could not be revived!
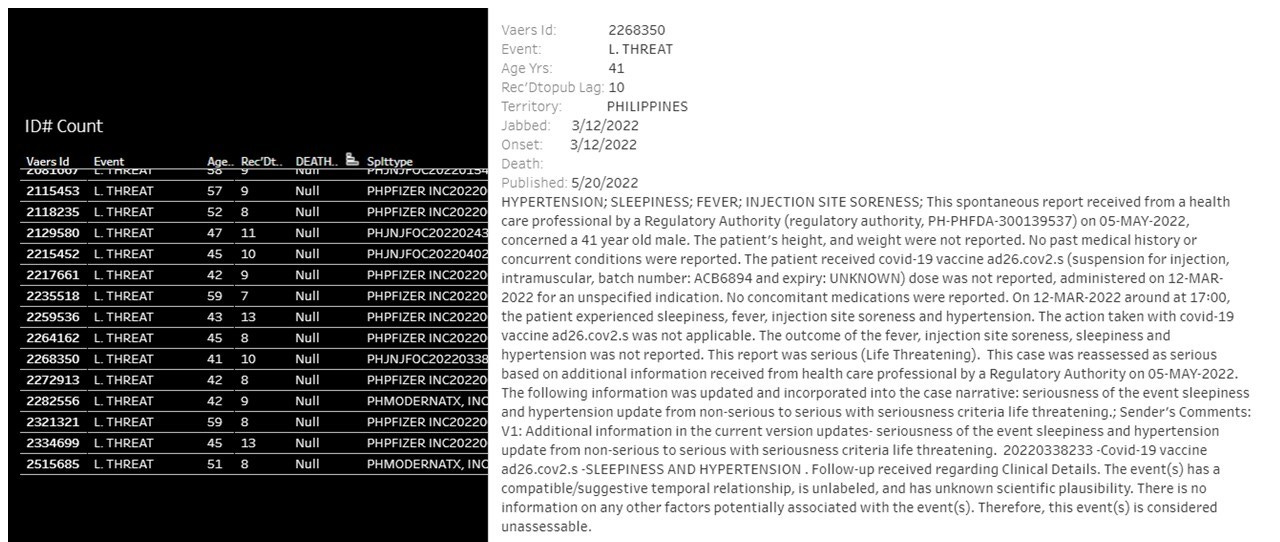
Hypertension and sleepiness immediately following receipt of Janssen dose ACB6894. Final outcome is not known.
Case of immediate anaphylaxis in 45-yo female, 20 minutes after injection with Janssen. The report stated that the patient recovered.
The VAERS reports of deaths (275) are very much lower that the Philippines FDA reports of deaths (793) for this age-group. Likely at least in part to considerable reports of deaths being due to Coronavac and other non-US manufactured products.
Further evaluation and root cause analyses for these deaths is required.




Wow Sally...how do you keep your eyes straight after sifting through all this depressing data?
Thank you.