Dengvaxia Exposed the Filipino People to Unnecessary Risks and Harm. Doesn't This Very Same Argument, on a Far Greater Even More Devastating Scale, Apply to Covid-19 Vaccines?
Dengvaxia officials face charges of reckless imprudence resulting in multiple homicide, violation of Anti-torture Law (RA9745), violation of the Consumer Act (RA7394) and the Pharmacy Act (RA10918)!
Leachon’s expert testimony in the Dengvaxia Case is very well covered by Arlie O. Calalo.
Specifically, the article elaborates the argument that the manufacturer was aware of the risks:-
That Phase 3 trials had not been completed before FDA registration.
And, that hasty approval and conduct of mass vaccination exposed Filipinos to serious health risks.
What irony! Twilight zone irony!
Let us contrast this stance with Dr. Leachon’s stance on Covid-19 vaccines. Please note Dr. Leachon’s public stance was generally that of the DOH. The DOH employed influencers to help drive product acceptance and uptake. As an influencer, Dr. Leachon got much public coverage. I reference this coverage here.
The concerns with the Covid-19 vaccines include:-
Covid-19 vaccines that were rushed to market under operation warp speed.
They did not complete Phase 3 trials before rushed rollout to the entire population under Emergency Use Authorization starting on 1st March 2021.
To date, only Pfizer has, in June 2023, been given CPR status; all other brands are still under EUA. Those products cannot be used when they finally expire (6 month shelf lives were extended up to 18 months).
Did the Philippines FDA do Independent Evaluation of C19 Vaccines? Answer = They were not required to do this!
The Philippines FDA approved the EUA use of the Covid-19 vaccines on the basis of other international authorities doing the same. Excerpt from 22 December 2021 updated EAU for Pfizer. Note that the FDA Director General was granted power to implement reliance on established regulatory authorities. Latest EUA documents available here.
I have not found any evidence that the Philippines FDA has independently re-evaluated the C19 vaccines since their EUA approvals. They have not changed their dosing recommendations, other than related to frequency of dosing and use of heterologous dosing, since the products were EUA authorized. If any of my readers has seen anything different, please do comment below.
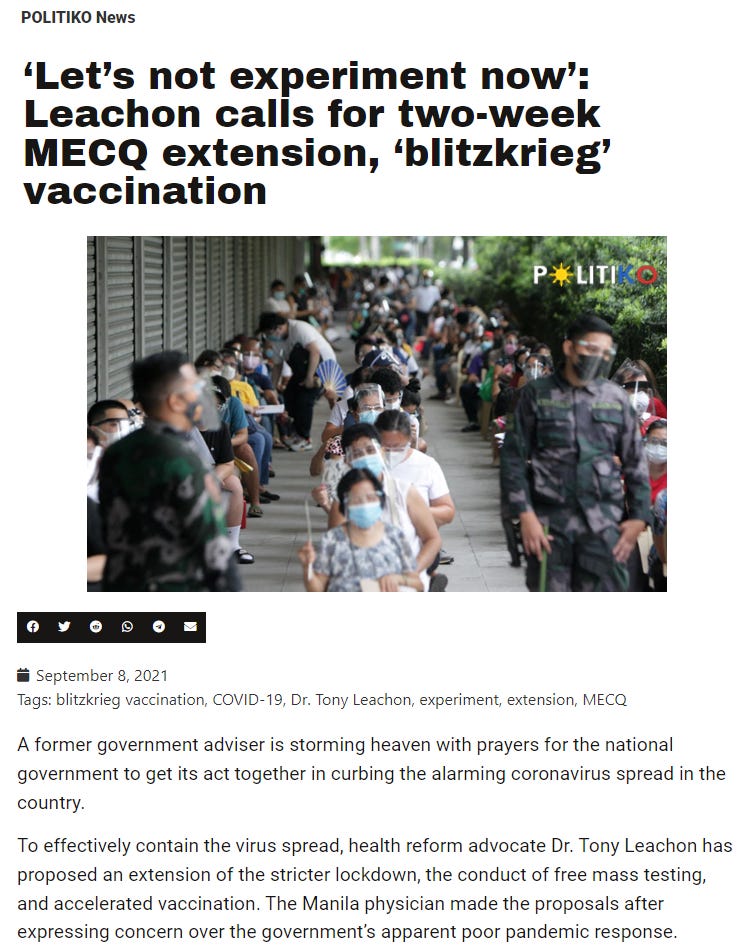
While Dr. Leachon questioned safety of Chinese-made Sinovac vaccines in December 2020,
by September 2021 he was pushing for “blitzkrieg vaccination” and extension of strict lockdowns.
He has been actively promoting Covid-19 vaccination and pandemic measures ever since, unreservedly pushing for mandatory 2nd booster dosing of Covid-19 vaccines to young adults as late as April 2023.
Dr. Leachon is now a Special Advisor to DOH against non-communicable diseases. He recommends Covid-19 vaccines for people with heart disease.
There are already serious questions from the likes of Drs. Malhotra and McCullough, among many others, about cardiac sequelae from C19 injections. The latest VAERS Data via openvaers shows an astounding number of reports of ill health following Covid-19 vaccination. Some of these conditions have been considered “non-communicable”. Will Dr. Leachon, in his role as special advisor, be dealing with the non-communicable diseases fallout from the very Covid-19 vaccination he and his DOH colleagues have been relentlessly pushing?
In the Dengvaxia case officials are facing multiple criminal charges.
Shouldn’t the same standards, rules, laws and precendent be used for the case of Covid-19?
















Pinoys were bitchin' about Dengvaxxia when it came out, but took the covid shot hook, line, and sinker. Lechon, yes, he is a pig, and needs his license pulled for his lies during the pandemic. I can't prove this, this asshole made money of the pandemic. Now, he's flipped?? I blasted him with facts and peer reviewed articles and he eventually blocked me on Facebook.
Easy. Leachon is a paid spokesperson on Pfizer's payola.
Sanofi didn't pay him so he spoke against Dengvaxia.
Sinovac didn't pay him so he spoke against the Sinovac COVID vaccine.
Only Pfizer is approved for minors in the Philippine COVID schedule so he advocated a "blitzkreig" of vaccinations for minors.
100% of shills agree with whatever harmaceutical is funding them :D
His posturing of concern for further study, taking a cautious approach, etc only serve his pocket. His statements happening to align with good medical and scientific practice are mere coincidence. Great if the science aligns with his pocket. If not, make up another reason why his pocket's needs override public health.